Australian Bragg Centre
South Australia

About
The Australian Bragg Centre is a purpose built biomedical development set to be home to leading cancer research institutes as well as the first clinically dedicated proton beam precision therapy centre in the Southern Hemisphere.
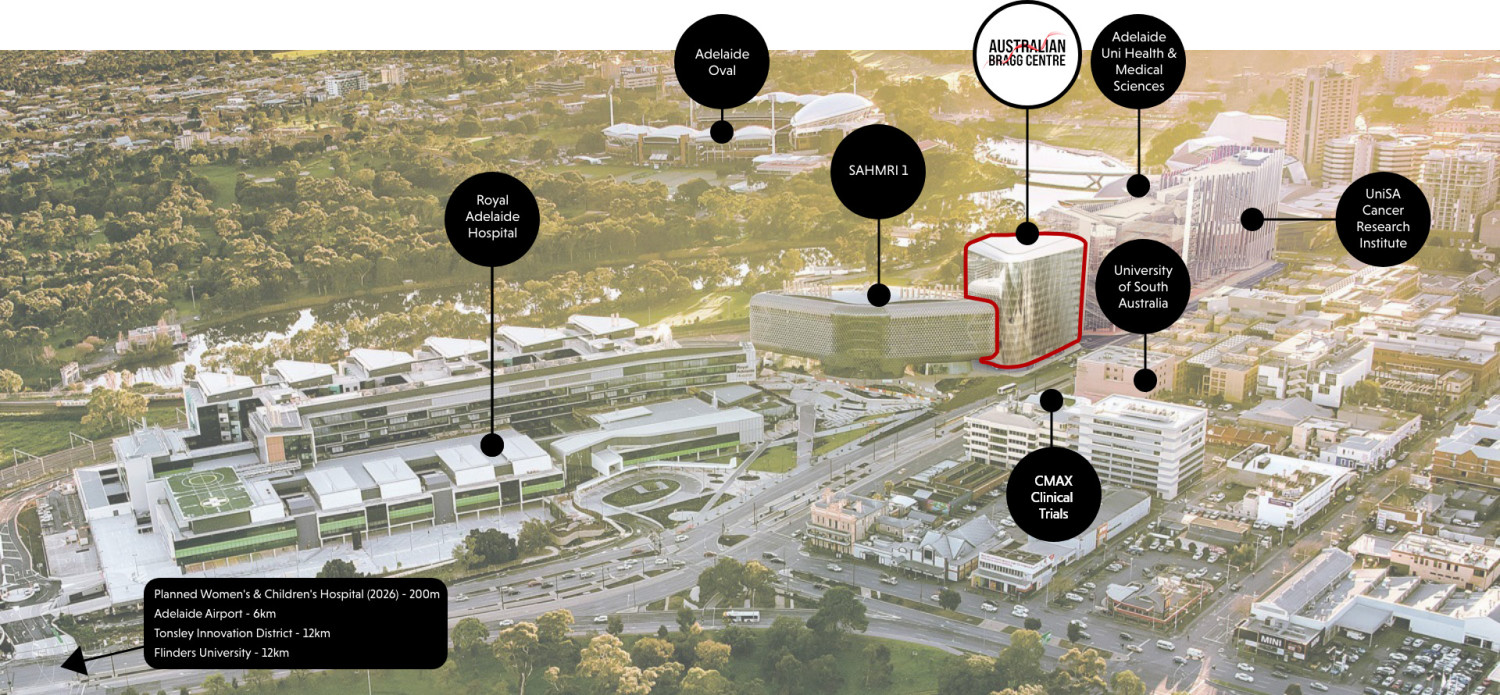
Located in the heart of Adelaide, the Australian Bragg Centre provides unmatched accessibility to a world-class, highly connected health and life sciences precinct that links with the most complete clinical trial ecosystem in Australia, world-leading AI and data analytics capabilities and a thriving cluster of medtech companies.
With limited floors remaining, a rare opportunity exists to secure the last remaining commercial space in Adelaide BioMed City | Health Innovation and Translation District until 2028.
Collaborate, innovate and inspire.
Adelaide biomed city
The $3.8B Adelaide BioMed City Health Innovation and Translation District is a collective initiative to bring together leading researchers, medical professionals, and industry leaders to collaborate on cutting-edge projects in areas such as genomics, precision medicine, and digital health.
The district features state-of-the-art facilities for research and development, as well as opportunities for start-ups and established companies to commercialise their innovations.
With a mission to deliver health and economic impact, locally and globally the district acts as a gateway for industry research collaboration, and a catalyst for commercialisation and improved patient outcomes.
The Australian Bragg Centre, situated between the South Australian Medical Research Institute and a stones throw from the Royal Adelaide Hospital, will play a pivotal role in the future growth and capabilities of the district.

Why Adelaide? You're in good company.

















UniSA City West Campus
UniSA is home to several prominent institutes and centres that conduct cutting-edge research in various areas, including health and wellness, rehabilitation, and chronic disease management, turning discoveries into solutions that benefit the economy and society. UniSA's approach to research is strongly aligned with industry, engaging in partnerships with business, industry and community, who help to shape the questions we ask.
UniSA's medical research programs provide students with the opportunity to gain hands-on experience in the field and work alongside leading researchers and practitioners. With its focus on innovation and impact, UniSA is playing a key role in advancing medical research in Australia and beyond.

The University of Adelaide
The University of Adelaide is one of Australia's oldest and most prestigious universities and ranked in the top 100 of universities worldwide.
The university has a long history of conducting groundbreaking research in the field of health sciences, and is home to several prominent research institutes, including the Robinson Research Institute and the South Australian Health and Medical Research Institute. These institutes conduct cutting-edge research in a range of areas, including reproductive health, infectious diseases, and cancer.
In addition to its research institutes, Adelaide University also offers a range of medical and health science programs, including undergraduate and graduate degrees in medicine, nursing, and public health. These programs provide students with the opportunity to gain hands-on experience in the field, and to work alongside leading researchers and practitioners.

Lot Fourteen
Lot Fourteen is a $2.2B world-class innovation district dedicated to driving productivity in emerging industries and solving complex global challenges. Home to more than 1500 people and 150 businesses, the dynamic community will reach 6000 by 2028.
More About Lot Fourteen >
Lot Fourteen Directory >
Entrepreneur and Innovation Centre and Innovation Hub >

Australian Bragg Centre
The Australian Bragg Centre is a purpose built bio-medical development set to be home to leading cancer research institutes as well as the first clinically dedicated proton beam precision therapy centre in the Southern Hemisphere.
The Australian Bragg Centre for Proton Therapy and Research will occupy the ground floor and three below-ground levels of the Australian Bragg Centre building on the eastern side of the distinctive SAHMRI building on North Terrace, Adelaide.

SAHMRI
SAHMRI is South Australia’s independent not-for-profit health and medical research institute.
We believe in a healthier future for all Australians. We listen to the needs of our communities to deliver research that changes the way healthcare is delivered. Through research excellence, innovation and strategic partnerships, we turn discoveries in health and medical research into impact for the community to improve their lives and the health of all Australians.
The creation of SAHMRI was a powerful investment in the health and quality of life for people throughout South Australia and beyond. SAHMRI and its partners continue to invest in a healthier future for all, including construction of the Australian Bragg Centre for Proton Therapy and Research on a site next to SAHMRI’s headquarters. This new building will house Australia’s first proton therapy unit, and the first of its kind in the Southern Hemisphere.

Royal Adelaide Hospital
The Royal Adelaide Hospital (RAH) is one of Australia's leading public hospitals. Providing excellence in patient care, cutting-edge technology, and innovative research programs, the RAH offers a range of services, including emergency care, surgery, and specialist medical services. With its state-of-the-art facilities and world-class expertise, RAH is a vital part of Adelaide's health system, and plays a crucial role in improving the health and well-being of the South Australian community.

The University of Adelaide Health & Medical Sciences Building
The University of Adelaide Health & Medical Sciences is a leading provider of health and medical education in Australia, seeking to improve health care in Australia and internationally.
The Faculty of Health and Medical Sciences offers a range of undergraduate and graduate programs in health sciences, including medicine, nursing, and public health, as well as research-focused programs in areas such as biomedical science and medical imaging. The Faculty is renowned for its world-class research, with a strong focus on areas such as infectious diseases, cancer, and reproductive health. By providing students with a rigorous education and opportunities for hands-on experience, the University of Adelaide Health & Medical Sciences is preparing the next generation of health and medical professionals to make a real difference in the world.

South Australian immunoGENomics Cancer Institute (SAiGENCI)
The Adelaide Health and Medical Sciences building is also home to the South Australian immunoGENomics Cancer Institute (SAiGENCI) which is a world-class cancer research institute, jointly resourced by the Federal Department of Health, CALHN and the University of Adelaide.

University of South Australia - Bradley Building (home to UniSA Cancer Research Institute)
UniSA is of one the world’s top young universities, globally connected and engaged with industry, collaborating with more than 2,500 partners worldwide. UniSA create knowledge that is central to global economic and social prosperity through education, research and partnership excellence. The Bradley Building is a leading destination for health research and teaching – and home to one of the most significant concentrations of cancer researchers in the state.

CMAX Clinical Research
CMAX is one of Australia’s largest and most experienced Phase I-II clinical trial units. Centrally positioned in Adelaide, South Australia and operating for more than two decades. CMAX is located opposite the Royal Adelaide Hospital and adjacent to the innovative Adelaide BioMed City precinct.
CMAX conducts successful world-leading research for local and international clients, specialising in a range of early-phase trials and first-time-in-human studies (FTIH). Since 1993 we have designed and delivered more than 800 early phase clinical trials, including more than 200 first in human studies for pharmaceutical, biotechnology and device companies.
We are proud that trials undertaken at CMAX, have resulted in the advancement of global medicines and technologies, from cancer treatment and pain control to early warning bio-devices and hormone replacement therapies.

Adelaide Oval
Adelaide Oval is a world-renowned cricket and football stadium located in North Adelaide. It has a seating capacity of over 53,000 and has hosted many major sporting events and concerts.

Festival Theatre
The Adelaide Festival Centre is a performing arts venue located in Adelaide's cultural precinct. It hosts a variety of local and international performances, including theatre, dance, music, and comedy, and is home to several resident companies.

Adelaide Railway Station
Adelaide Railway Station is the main railway station in Adelaide. It serves as a hub for interstate and regional train services and is a historic landmark.

Rundle Mall
Rundle Mall is a popular shopping precinct located in the heart of Adelaide. It features over 700 retailers, including major department stores, boutique shops, eateries, and is a popular destination for both locals and tourists.

River Torrens Footbridge
The Adelaide Footbridge is a pedestrian bridge that spans the River Torrens in Adelaide. It connects the Festival Centre on the northern bank to the Adelaide Oval on the southern bank and offers stunning views of the city skyline.

SKYCity Adelaide
A popular entertainment destination located in the heart of Adelaide. It features a range of gaming options, including table games and electronic gaming machines, as well as several bars and restaurants.

Adelaide Convention Centre
The Adelaide Convention Centre is a premier conference and events venue located in the heart of Adelaide. It offers state-of-the-art facilities and hosts a variety of local and international events.

Situated in Adelaide's physical and intellectual ‘innovation belt’, the Australian Bragg Centre offers lifestyle, amenities and flexible transit options.
Opportunity
Space that inspires.
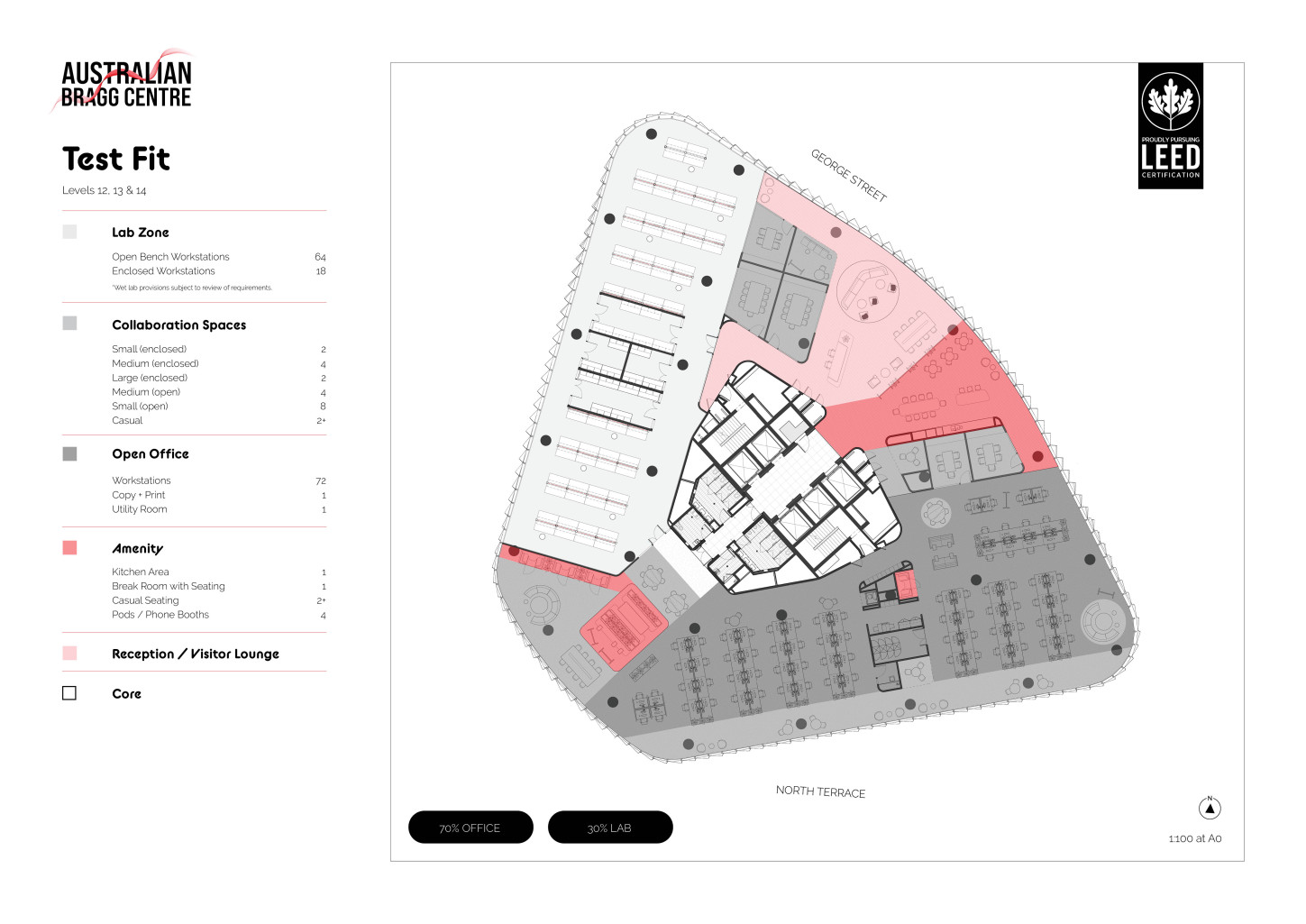
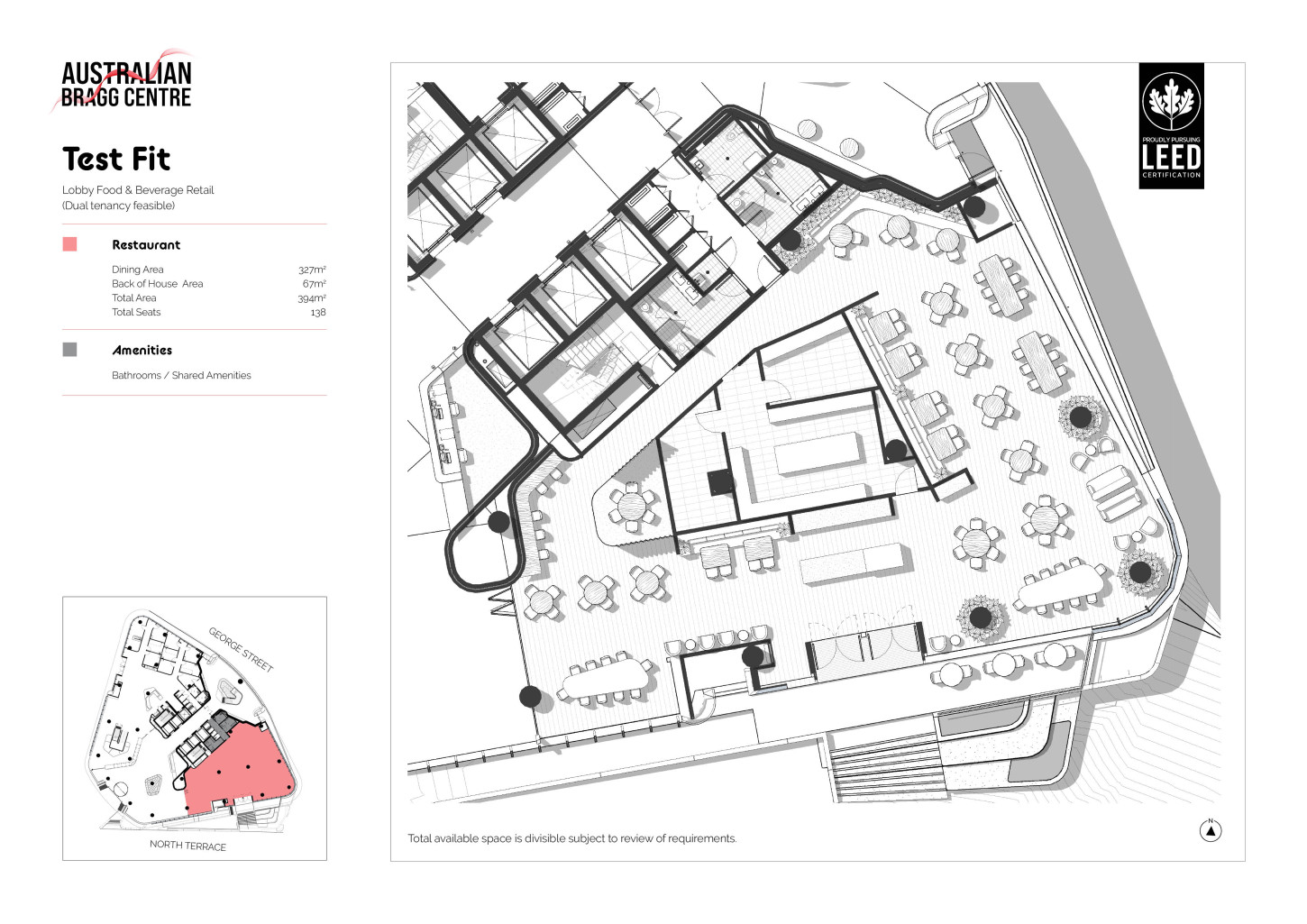
The cutting edge design of the Australian Bragg Centre offers an energising environment designed and created by Australia’s leading development team. Going above ‘fit for purpose’ the spaces are created with end-user in mind, allowing flexibility for growth, adaptive use and enabled for research and development.
Share artfully designed spaces, tools and technology with some of Australia’s greatest minds in cancer research. Access talent, resources, investment, and innovation practices all underpinned by community and amenities.
Sustainability infused design and amenities work to support a healthy and productive workplace environment that is anticipated to achieve LEED Gold standard.
Wet lab accommodation (subject to negotiation)



Team
Cutting edge project team.
Brought to you by an award-winning team
committed to exceeding expectations.
Established in 1997, Commercial and General (C&G) is South Australia’s most prominent developer with $2.2 billion in projects delivered or under development. With a dedicated healthcare practice group, C&G have specialised expertise and an unmatched ability to deliver life science developments of scale. The C&G project portfolio includes the successful delivery of the $345 million Calvary Adelaide Hospital, the award-winning $1b WEST redevelopment of Football Park, and the $250m Eighty Eight O’Connell mixed-use development being built in North Adelaide.